Hydrogen Fuel Cell Cars
In addition to fuel economy and car pollution, hydrogen fuel cell cars have a few other paybacks as well.
Lower repair costs from fewer moving parts, a longer driving range, the ride is quieter and smoother.
There is plenty of power for devices like GPS, DVD movie players and Internet connections.
The distinction between hydrogen fuel cells and batteries is that batteries sooner or later go dead. A hydrogen fuel cell
stays alive as long as hydrogen and oxygen are introduced to the cell, producing electricity continuously.

There are quite a few different types of fuel cells and each uses a different chemistry.
They are typically distinguished by electrolyte type and operating temperature.
Fuel cells provide a DC current that can be used to power lights, motors and an array of other electrical devices.
Most are not suitable for cars but for larger stationary power generators.
The PEMFC (polymer exchange membrane fuel cell), also known as Proton Exchange Membrane, is the one most appropriate for cars
and is also what the Department of Energy is planning on using in the future to power our vehicles.
The PEM fuel cell uses one of the simplest reactions of any fuel cell. It is also shows the most potential for powering
vehicles and maybe even our homes in the future.
How Hydrogen Fuel Cell Cars Work
They work by using hydrogen fuel and oxygen from the air to create electricity.
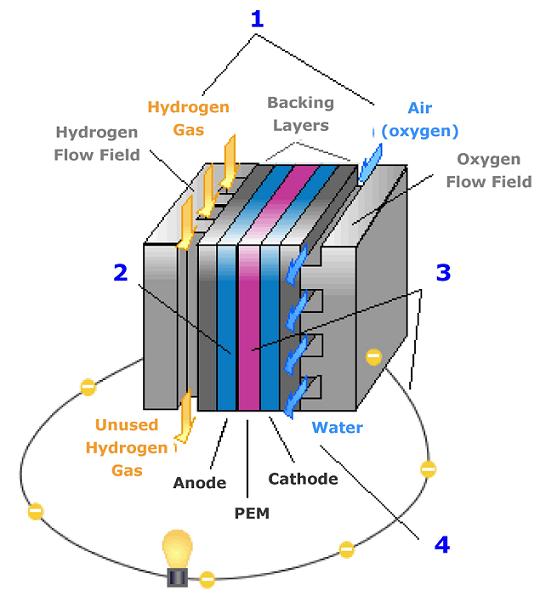 PEM Fuel Cell 1-Hydrogen fuel is channeled through field flow plates to the anode on one side of the fuel cell, while oxygen from
the air is channeled to the cathode on the other side of the cell.
2 - At the anode, a platinum catalyst causes the hydrogen to split into positive hydrogen ions (protons) and negatively charged electrons.
3 - The polymer Electrolyte Membrane (PEM) allows only the positively charged ions to pass through it to the cathode.
The negatively charged electrons must travel along an external circuit to the cathode, creating an electrical current.
4 - At the cathode, the electrons and positively charged hydrogen ions combine with oxygen for form water, which flows out of the cell.
Most fuel cells designed for use in vehicles produce less than 1.16 volts of electricity, not even close enough
to power a vehicle. So, multiple cells must be assembled together to make a fuel cell stack. The potential power
generated by a fuel cell stack will depend on the quantity and size of each fuel cell that consists of the stack and
the surface area of the PEMFC.
Fuel Cell Cars Efficiency
The type of hydrogen fuel that is used is the main factor in its efficiency. If it isn't pure hydrogen, then the vehicle will need
a reformer, which turns the hydrocarbon or alcohol fuels into hydrogen. A reformer produces heat a creates other gases along with the hydrogen.
This lowers the efficiency of the fuel cell.
When pure hydrogen is used the efficiency goes up. Hydrogen fuel cell cars have the potential to be approximately 65% efficient.
|